Engineered for the World’s most Regulated Industry
From APIs to packaging, pharmaceutical supply chains are complex. Rising disclosure norms mean buyers, regulators, and investors expect verifiable, audit-ready, batch-specific carbon data.
Book a Demo
Real impact starts with Batch-level Clarity
APIs, intermediates, and packaging span complex supply chains. Without supplier data, Scope 3 footprints stay incomplete and miss key hotspots.

Upstream Emissions are the biggest Blind Spot
Ingredient and process changes need manual recalculation. This slows sustainability teams and delays footprint reporting and disclosures.

Frequent Formulation changes Slow Reporting
Clients and investors now demand batch-level data to verify carbon reports. Companies lacking structured ESG data risk credibility loss.

Traceability from Molecule to Batch is now expected
Powering the Next Era of Sustainable Pharma
From sourcing to scale-up, track carbon across formulations, facilities, and suppliers. Ensure data stays traceable, audit-ready, and aligned with evolving disclosure norms, without extra workload.

Conduct Climate Risk Assessments across your Value Chain
Climate risk in pharma spans physical and transition factors. Mapping flood-prone sites, shifting policies, and investor demands helps build resilience into procurement and supplier planning.
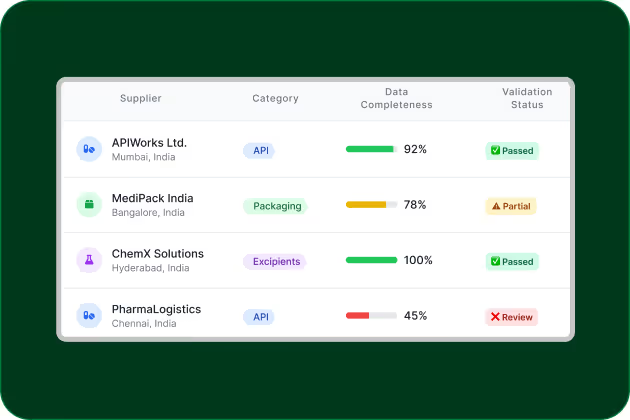
Build Supplier-level Visibility for credible Scope 3 data
Pharma supply chains are fragmented. Collecting consistent supplier data on energy, logistics, and materials enables audit-grade Scope 3 calculations and CDP-ready reporting.

Create Batch-level Product footprints using Real-world inputs
Accurate LCAs reflect batch-specific process variations. Generating cradle-to-gate data improves product-level reporting and strengthens buyer, regulator, and certification responses.

Set Science-Based Targets and Model Reduction Pathways
Switching solvents or improving formulation efficiency affects emissions differently. Scenario modelling aligns actions with SBTi guidance and builds science-based reduction roadmaps.

Map disclosures to CDP and Biodiversity-linked frameworks
As reporting expands to water, biodiversity, and supply chain impacts, pharma companies must stay ahead. Structured data aligned to CDP categories improves transparency and speeds up reviews.
The System behind Smarter Pharma Sustainability
Handling pharma emissions means batch variability, multi-site data, and strict formats. Our system unifies structured collection with intelligence for simpler, credible reporting.
Working with the Makers of Healthcare
We help pharmaceutical teams build verified, audit-ready sustainability data before it becomes a compliance challenge.
We work closely with:
Drug Discovery Firms: Gain upstream visibility across APIs and intermediates to close Scope 3 gaps and meet investor expectations
Medical Device Manufacturers: Trace emissions across production and operations with verified, compliance-ready data
CDMOs & Contract Partners: Manage multi-client cycles while delivering accurate, consistent sustainability metrics for buyers

For Every step of your Low-Carbon Journey

For your Customers
Pharma buyers and partners now expect verified emissions and batch-level data. Traceable information builds trust in audits, tenders, and supply contracts.

For your Company
Accurate data supports CDP, CSRD, and buyer needs without manual work. It guides sourcing, procurement, and product design decisions internally.

For the Planet
Pharma production drives emissions, water use, and biodiversity impact. Better visibility helps companies act faster and strengthen climate resilience.



.avif)








.avif)















.svg)
.svg)